Australia is currently muddling through a critical shortage of EpiPen Jr epinephrine auto-injectors, devices intended as an emergency treatment for young children who suffer anaphylaxis, a serious, sometimes life-threatening allergic reaction.
EpiPen brand auto-injectors have been in short supply globally due to manufacturing issues at Meridian Medical Technologies, a division of Pfizer that produces the devices. Meridian is currently the subject of a civil investigation by the US Attorney’s office for the District of NY.
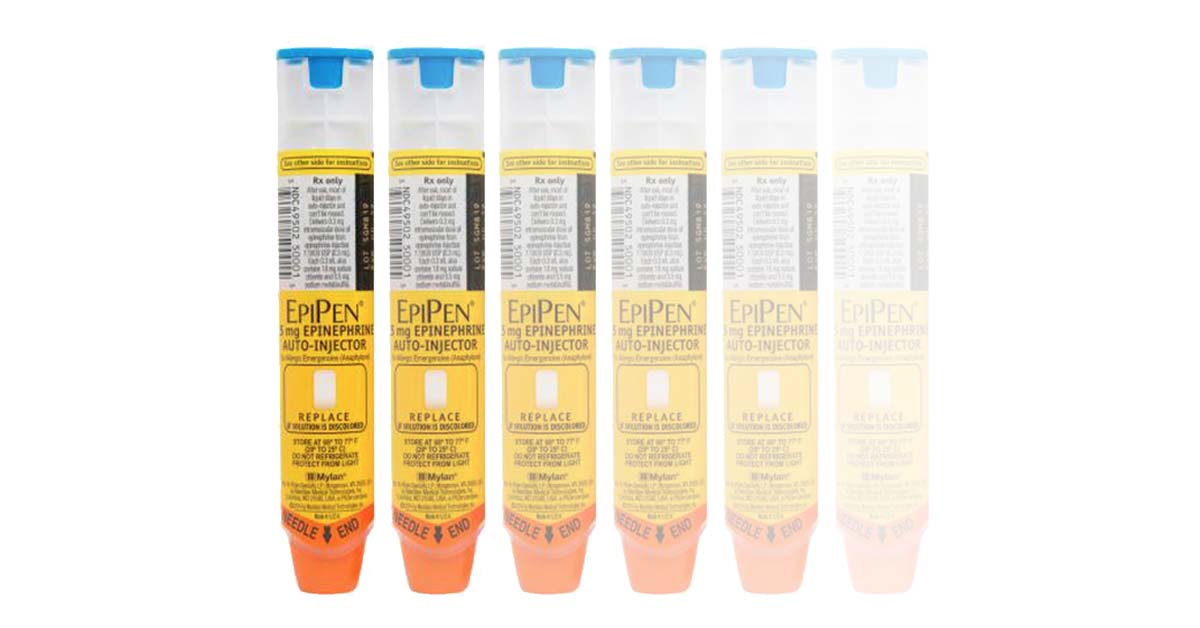
The shortage was so acute that the Therapeutic Goods Administration (TGA), the Australian agency responsible for regulating drugs, previously advised parents to use the devices past their expiration date.
Now, the TGA has approved a batch of the devices found to contain traces of pralidoxime. The agency says this was likely caused by a manufacturing issue between the production of different products, noting the manufacturer has taken action to prevent the issue for future batches.
“Pralidoxime is used to treat people who have been poisoned by pest-control chemicals and its safety profile has been well established in humans,” a spokesperson for the TGA says.
“In the situations in which EpiPen Jr is used, administration of adrenaline can be life-saving. The risk from not having adrenaline available to treat anaphylaxis is far greater than the risk of being exposed to a very small amount of pralidoxime.”
The agency expects the current shortage of EpiPen Jr to continue through January of next year.
Announcing the Holiday Edition of The Safe Snack Guide to Help You Plan a Safe and Inclusive Celebration!