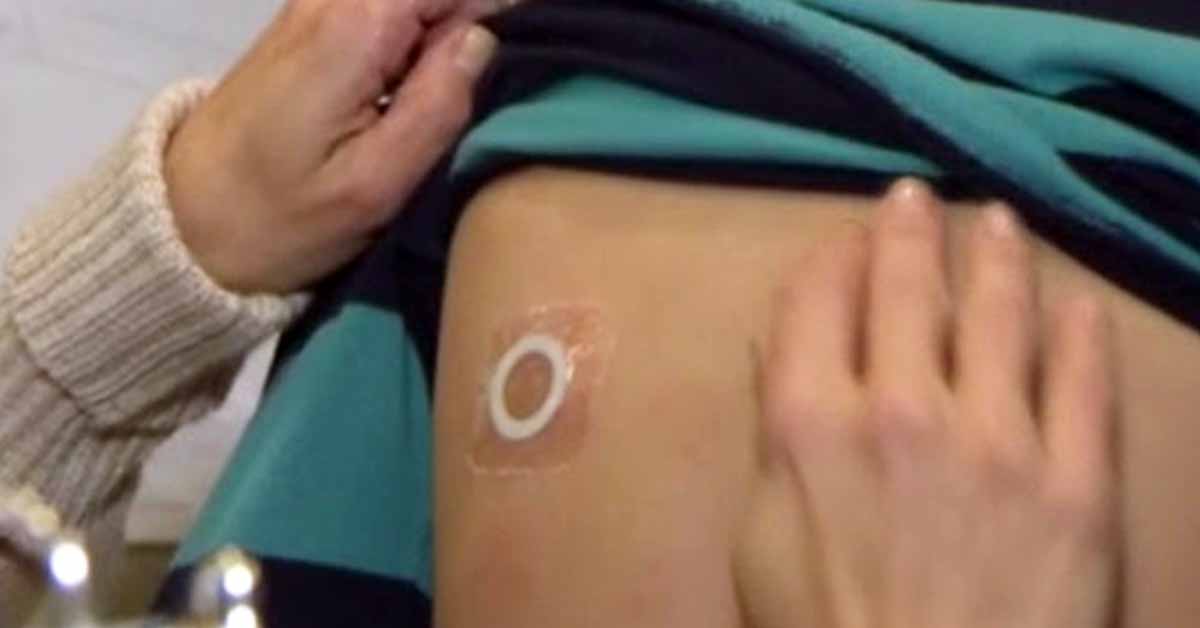
Two weeks ago, we reported that DBV Technologies was launching a Phase 3 clinical study of their redesigned Viaskin Peanut patch in 4-7 year-olds, a therapy intended to desensitize patients to their peanut allergy. DBV was forced to redesign the patch when the US Food and Drug Administration (FDA) voiced concerns about the patch’s adhesion in 2020.
Now the FDA has stepped in again to issue a partial hold on the trial, dubbed “VITESSE”. It is not known how long the FDA’s response will delay the VITESSE trial as DBV will be forced to
Below is the press release issued by DBV yesterday announcing the FDA’s action.
DBV Technologies Provides Clinical Update on VITESSE Phase 3 Trial
DBV Technologies (Euronext: DBV – ISIN: FR0010417345 – Nasdaq Stock Market: DBVT), a clinical-stage biopharmaceutical company, today announced that it received feedback from the U.S. Food and Drug Administration (FDA) in the form of a partial clinical hold on its VITESSE (Viaskin Peanut Immunotherapy Trial to Evaluate Safety, Simplicity and Efficacy) Phase 3 clinical study.
In the partial clinical hold letter, the FDA specifies changes to elements of the VITESSE protocol with the intent for the trial to support a future BLA submission. Within the FDA’s communication, the modifications address design elements, including the statistical analysis of adhesion, minimum daily wear time and technical alignments in methods of categorizing data, to meet study objectives as well as the total number of trial participants on active treatment.
DBV has not yet begun the screening or recruitment of subjects in the VITESSE study. The partial clinical hold is specific to VITESSE and does not impact any other ongoing DBV clinical studies. The Company expects to provide additional updates following consultation with the FDA.
“We are grateful for the FDA’s additional feedback reflecting careful attention to DBV’s VITESSE study,” said Daniel Tassé, Chief Executive Officer of DBV Technologies. “We are pleased that the comments to the protocol are specific, clear and arrived prior to enrollment. We look forward to upcoming discussions with key FDA personnel.”
VITESSE is a Phase 3, double-blind, placebo-controlled, randomized study to assess the efficacy and safety of epicutaneous immunotherapy with the modified Viaskin™ Peanut 250 µg patch in peanut-allergic children ages 4 to 7 years.
About DBV Technologies
DBV Technologies is developing Viaskin™, an investigational proprietary technology platform with broad potential applications in immunotherapy. Viaskin is based on epicutaneous immunotherapy, or EPIT™, and is DBV Technologies’ method of delivering biologically active compounds to the immune system through intact skin. With this new class of non-invasive product candidates, the Company is dedicated to safely transforming the care of food allergic patients. DBV Technologies’ food allergies programs include ongoing clinical trials of Viaskin Peanut. DBV Technologies has global headquarters in Montrouge, France, and North American operations in Basking Ridge, NJ. The Company’s ordinary shares are traded on segment B of Euronext Paris (Ticker: DBV, ISIN code: FR0010417345) and the Company’s ADSs (each representing one-half of one ordinary share) are traded on the Nasdaq Global Select Market (Ticker: DBVT).
Forward-Looking Statements
This press release may contain forward-looking statements and estimates, including statements regarding the therapeutic potential of Viaskin™ Peanut as a treatment for peanut-allergic children and the potential benefits of EPIT™, DBV Technologies’ clinical development and regulatory plans, timing and projections of VITESSE study milestones, and timing and anticipated results of interactions with regulatory agencies. All statements about VITESSE study milestones, enrollment and anticipated results contained herein are DBV’s best estimates and projections are based on performance of previous studies and are subject to known and unknown risks, uncertainties, and other factors that may cause actual results, performance and achievements with respect to the VITESSE study to differ materially from the estimates and projections contained herein. These forward-looking statements and estimates are not promises or guarantees and involve substantial risks and uncertainties and may be impacted by market conditions as well as other risks and uncertainties set forth in DBV Technologies’ regulatory filings with the Autorité des Marchés Financiers (“AMF”), DBV Technologies’ filings and reports with the U.S. Securities and Exchange Commission (“SEC”), and future filings and reports made with the AMF and SEC. Existing and prospective investors are cautioned not to place undue reliance on these forward-looking statements and estimates, which speak only as of the date hereof. Other than as required by applicable law, DBV Technologies undertakes no obligation to update or revise the information contained in this Press Release.
Investor Contact
Anne Pollak
DBV Technologies
+1 857-529-2363
anne.pollak@dbv-technologies.com
Media Contact
Angela Marcucci
DBV Technologies
+1 646-842-2393
angela.marcucci@dbv-technologies.com
Viaskin and EPIT are trademarks of DBV Technologies.