Important Update on EpiPen® (epinephrine injection, USP) 0.3 mg Auto-Injectors
Extended Expiration Dates for Select Lots of EpiPen® 0.3 mg AutoInjectors and Its Authorized Generic
To address shortages of EpiPen, Pfizer is coordinating with FDA to extend the expiration dates of specific lots of EpiPen® 0.3 mg Auto-Injectors and its authorized generic version after review of stability data. Patients should have confidence in using the products from these particular lots as Pfizer works to stabilize supply, which is anticipated in the fourth-quarter of 2018.
This announcement is based on a careful review of product stability data provided by Pfizer. We believe the extension of the expiration date will temporarily address patients’ access to and use of EpiPen® 0.3 mg Auto-Injectors, and the authorized generic, particularly during back-to-school season as demand increases.
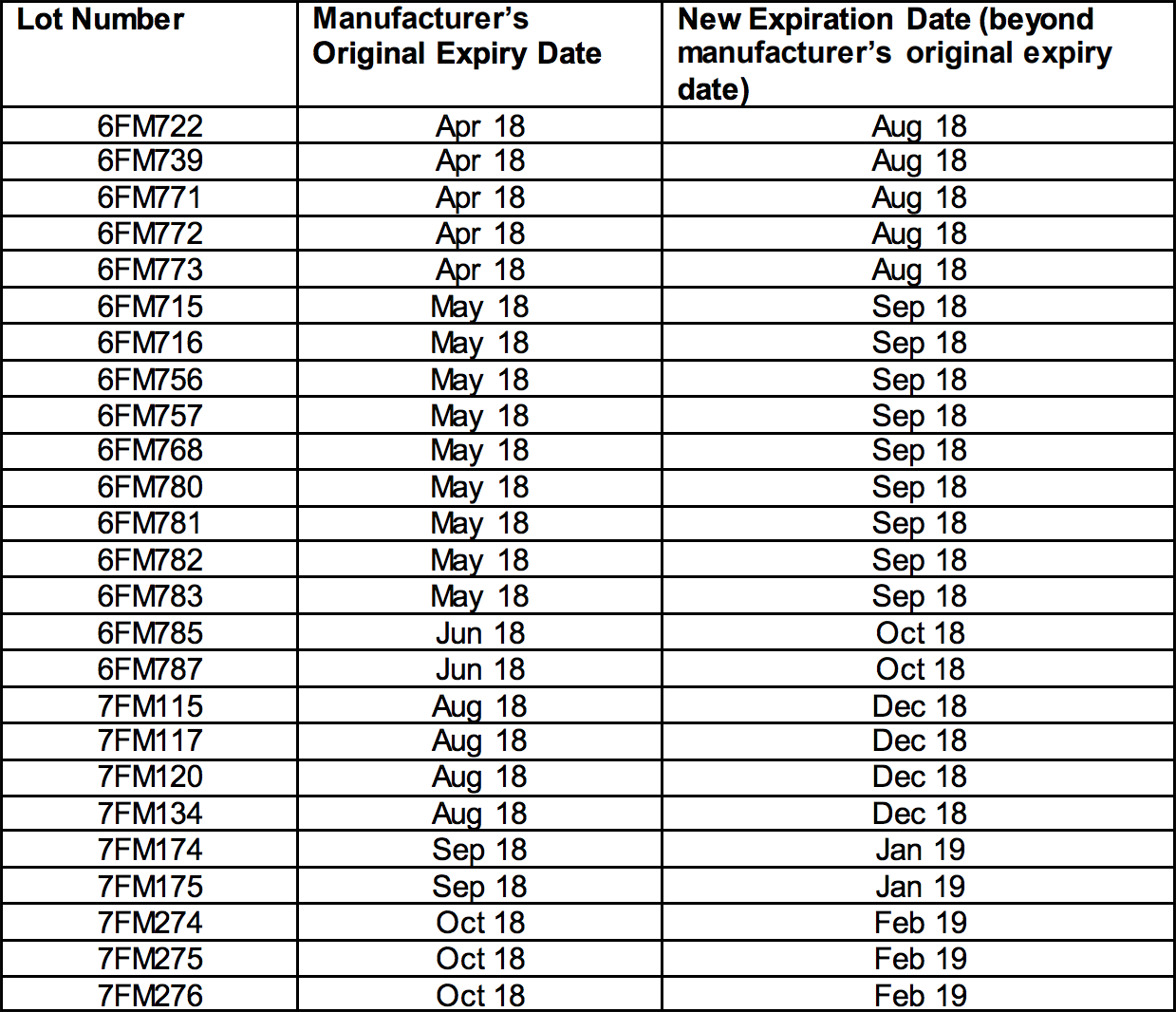
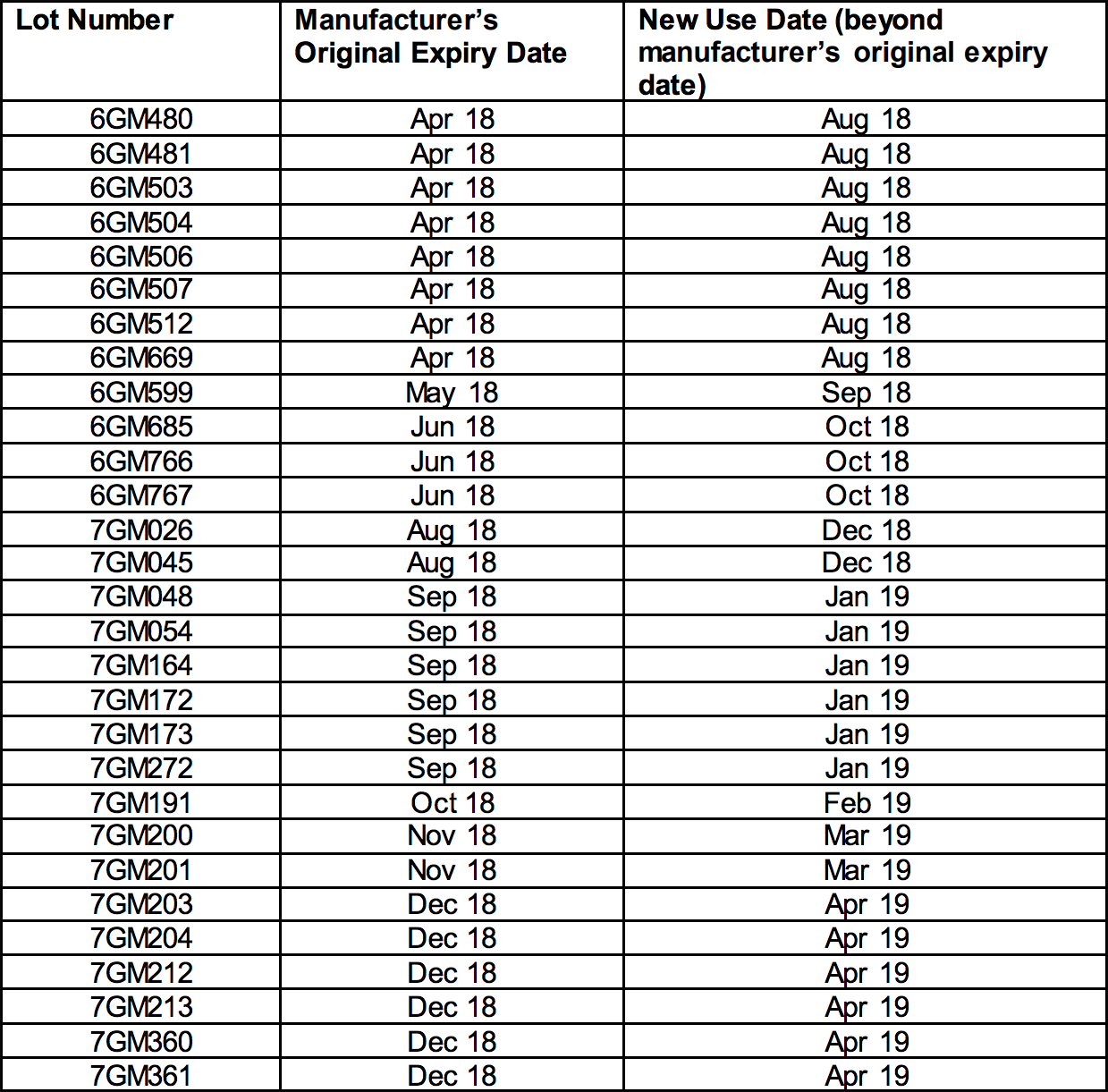
The affected lots, which have current expiration dates between April 2018 and December 2018, are indicated in the tables below with their new expiration dates and can be found on FDA’s website.
The extension of the expiration dates does not apply to EpiPen Jr® (epinephrine injection, USP) 0.15 mg Auto-Injectors and its authorized generic version of this strength. Patients should continue to adhere to the manufacturer’s expiry date labeled on EpiPen Jr® 0.15 mg and Epinephrine Injection, USP Auto-Injectors 0.15 mg products.
Please watch this important video from Pfizer’s Chief Medical Officer, Dr. Freda Lewis Hall, regarding this announcement:
Meridian, a subsidiary of Pfizer that manufactures EpiPen® 0.3 mg and EpiPen Jr® 0.15 mg Auto-Injectors, and the authorized generic versions of these strengths, continues to experience manufacturing constraints affecting supply. Mylan and Pfizer place great importance on the consistent availability of EpiPen for everyone who needs it. Pfizer is working tirelessly to increase production and expedite shipments as rapidly as possible. Currently, supplies will vary from pharmacy to pharmacy, and we cannot guarantee that product will be available at all pharmacies.
We understand the challenges this situation continues to pose to patients. Ensuring continuity of the supply of our medicines is paramount.
Mylan, the distributor, is expediting shipments upon receipt from Pfizer and continues to encourage patients who are experiencing difficulty accessing product to call its Customer Relations team at 800-796-9526 for assistance in locating other pharmacies. To further assist patients during this period of limited supply, Mylan recently extended its Customer Relations hours to Monday through Friday, 8 a.m. to 7 p.m. ET and Saturday through Sunday, 11 a.m. to 4 p.m. ET.
Epinephrine Injection, USP Auto-Injectors 0.3 mg
NDC 49502-102-02 appears on the box
NDC 49502-102-01 appears on the individual device within the box
EpiPen® (epinephrine injection, USP) 0.3 mg Auto-Injectors
NDC 49502-500-02 appears on the box
NDC 49502-500-01 appears on the individual device within the box





Been saying this for years! They last longer than we’ve been told. Funny how fast Pfizer came up with that data. It’s like they already had it or something…..