There’s some exciting news to report regarding innovations in auto-injector design, especially welcome in the wake of the recent Auvi-Q and Allerject recalls by Sanofi.
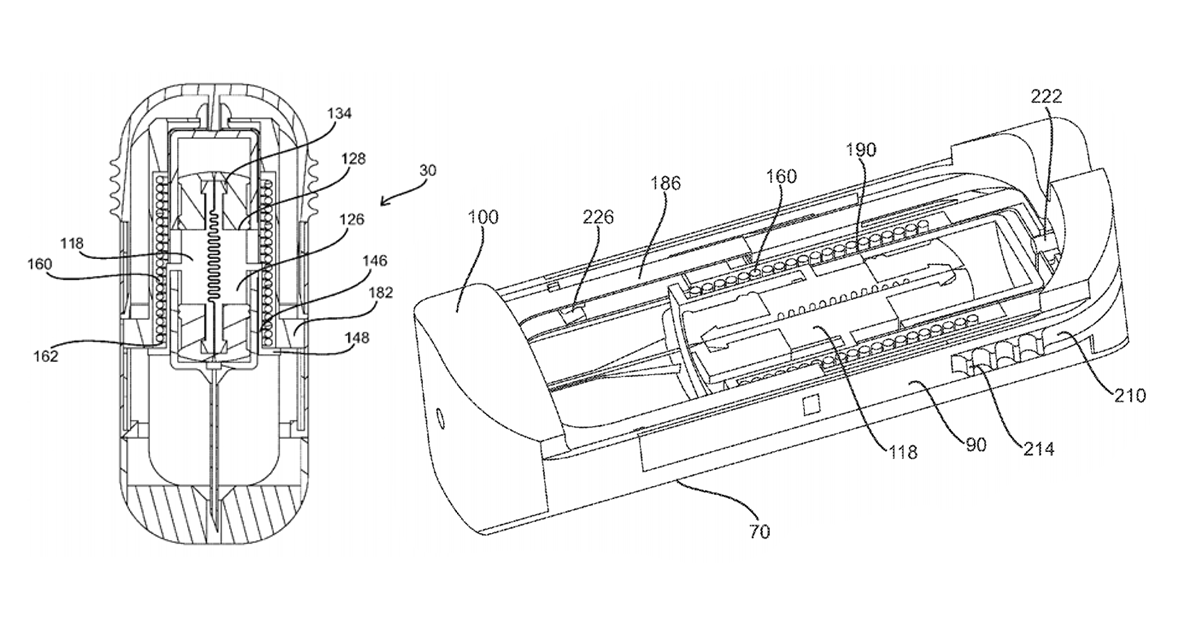
US Patent Number 9199037, entitled “Portable drug mixing and delivery system and method“, was just assigned to Windgap Medical, Inc, based in Somerville, MA. In a nutshell, the patent describes a multi-chambered mechanism with separate containers for wet and dry ingredients that mixes the components upon activation, just before the drug is administered.
The idea is to produce an auto-injector where the active medication (in this case, epinephrine) is kept dry until the moment it is needed. A dry form of the medication would conceivably be less prone to degradation due to age and temperature variations.
Here’s the opening statement summarizing the invention:
SUMMARY OF THE INVENTION
It has been recognized that if a drug can be kept out of the liquid phase and stored as a dry medication, the shelf-life, temperature susceptibility may increase allowing the efficacy and potency of the drug to endure longer and through harsher environments.
It has been recognized that a smaller drug delivery device than a conventional epinephrine auto-injector, which could be attached to a key chain and/or easily fit in a person’s pocket, would make the device easier to carry and more likely that the user will have it on their person when needed. An example of such a device package, purely for the purpose of comparison, could be sized similarly to that of a USB “thumb drive” which is designed to be with users on a fairly constant basis. For example, an auto-injector device embodiment has dimensions of 3 inches by 1 inch by ½ inch. However, dimensions of an auto-injector device may vary.
Does this patent really portend an auto-injector with a longer shelf-life, impervious to temperature changes, and the size of a USB thumb-drive that could be carried on a key chain?
We’ve reached out to the CEO of Windgap Medical and hope to have more information soon. Stay tuned!





I would guess the answer to at least the questions “impervious to temperature change” would be no. You still would need the liquid to be in liquid form. You can’t mix ice with a powdered medication and inject it. Longer shelf life and high temperature extreme perhaps.