The global food allergy market is entering a period of significant change, evolving beyond a longstanding focus on strict avoidance and emergency management toward the development of targeted therapies, including immunotherapies and biologics. According to an analysis by DelveInsight, the market across the seven major markets (7MM)—the United States, Germany, France, Italy, Spain, the United Kingdom, and Japan—is projected to grow from approximately $3.5 billion in 2025 to about $9.2 billion by 2034.
The United States remains the primary driver of this growth, accounting for the largest share of the market. In 2024, the US market was valued at roughly $2.4 billion, representing nearly 79% of the total 7MM market. This leadership is attributed to higher diagnosis rates and greater adoption of newer therapies, a trend expected to continue through the forecast period.
Epidemiological data underscores the scale of the challenge. Approximately 60 million diagnosed cases of food allergy were reported across the 7MM in 2024, including about 36 million in the United States alone. The report highlights multiple contributing factors, noting that sensitization through the skin and conditions such as atopic eczema in infancy are associated with increased risk. This growing patient population continues to drive demand for more effective long-term management options.
Epinephrine remains the cornerstone of emergency treatment for anaphylaxis, but it does not address the underlying immune response. As DelveInsight notes, traditional management strategies centered on avoidance and emergency intervention “do not target the root immune mechanisms and offer no long-term control,” underscoring the need for disease-modifying approaches.
The introduction of PALFORZIA (peanut allergen powder-dnfp), the first FDA-approved oral immunotherapy (OIT) for peanut allergy, marked an important step forward. At the same time, the report notes that uptake had been gradual before the drug was discontinued, and attention is shifting toward additional approaches such as epicutaneous immunotherapy (EPIT), including DBV Technologies’ Viaskin Peanut, which may offer a noninvasive alternative if approved.
A key regulatory milestone came in March 2025 with the FDA approval of OMLYCLO (omalizumab-igec), the first interchangeable biosimilar to Xolair for IgE-mediated food allergies. This development is expected to expand treatment options and increase competition within the biologics space.
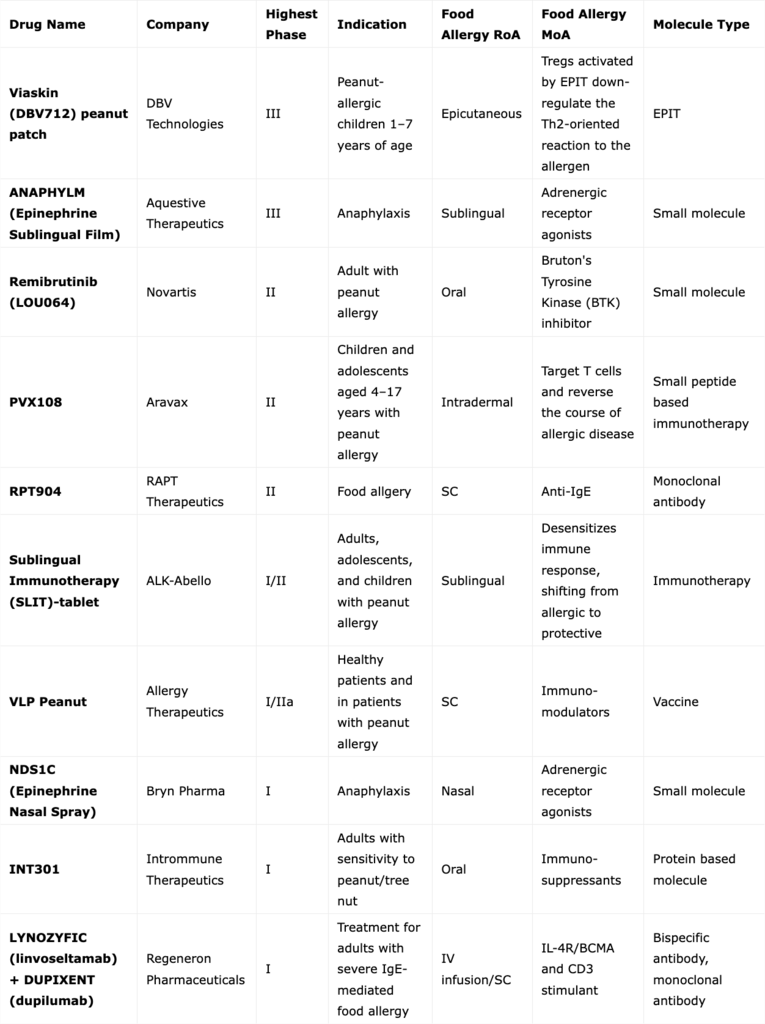
The treatment pipeline remains active, with multiple companies advancing therapies across different mechanisms of action. In addition to DBV Technologies, companies such as Novartis, Aravax, ALK-Abelló, InnoUp Farma, and Aquestive are contributing to a growing landscape of potential new treatments aimed at improving long-term outcomes.
Adult food allergy represents a significant unmet need. In 2024, severe cases among adults in the 7MM totaled nearly 25 million, exceeding mild-to-moderate cases. Despite this, treatment options for adults remain limited, highlighting a substantial opportunity for future therapeutic development, including candidates such as remibrutinib.
Allergen-specific trends also shape the market. While early innovation focused heavily on peanut allergy, the field is expanding toward broader treatment strategies, including therapies that may address multiple allergens.
Challenges remain, particularly in diagnosis. The report notes that overlapping symptoms and limitations in current testing methods can lead to underdiagnosis or misdiagnosis, complicating effective management and treatment selection.
Looking ahead, continued investment in new therapies and evolving treatment approaches is expected to reshape the market. With a projected compound annual growth rate (CAGR) of 11.30%, the food allergy market is poised for substantial expansion over the next decade.
Food Allergy Drugs Currently Under Development
In summary, the field is gradually shifting from a reactive model centered on avoidance and emergency care toward a more proactive approach that seeks to improve long-term outcomes. Growth will be driven by rising prevalence, expanding therapeutic options, and increasing focus on unmet needs across both pediatric and adult populations.
For families managing food allergies, these developments signal cautious but meaningful progress. For decades, the standard approach has centered on strict avoidance and carrying epinephrine for emergencies. While those measures remain essential, the growing pipeline of treatments suggests that more proactive options may become available.
That said, most of these therapies are still emerging, and access, cost, and long-term effectiveness will be key factors in how widely they are adopted. Treatments like oral and epicutaneous immunotherapy aim to reduce sensitivity to allergens, but they are not cures and may not be suitable for everyone.
The expansion of treatment options—particularly for adults, who have historically had fewer choices—is an encouraging sign. At the same time, accurate diagnosis remains critical, as misdiagnosis or incomplete evaluation can lead to unnecessary restrictions or missed risks.
For now, families should continue to rely on proven safety strategies: careful label reading, avoiding known allergens, and carrying epinephrine at all times. But the broader shift in research and development offers hope that managing food allergies in the future may involve more than just avoidance—it may increasingly include treatment aimed at reducing risk and improving quality of life.