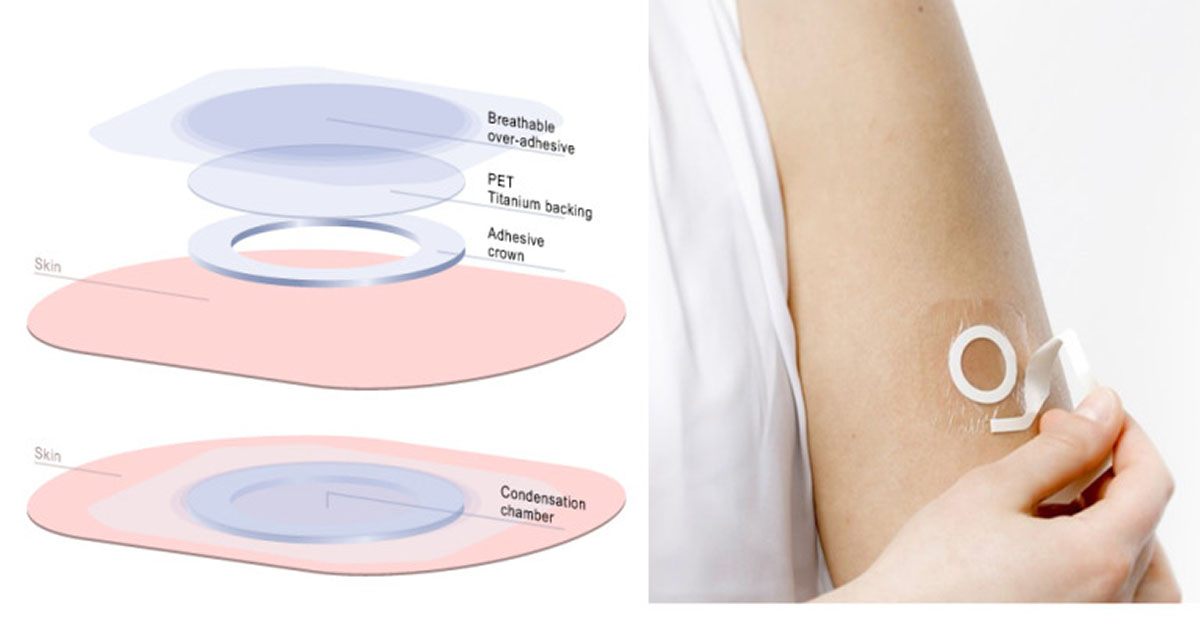
Presenting at the annual meeting of the American Academy of Allergy, Asthma & Immunology (AAAAI) in Atlanta on Monday, DBV Technologies, the pharmaceutical company developing the Viaskin family of “patch” therapies, announced the results of the extension to its Phase IIb clinical trial of its Viaskin Peanut therapy.
Dr Wayne Shreffler, Division of Allergy and Immunology, Department of Pediatrics, Massachusetts General Hospital, Harvard Medical School and investigator of the study, reported that 83.3% of pediatric patients responded to Viaskin Peanut 250µg after three years with a favorable safety profile and no serious adverse effects (SAEs) related to treatment.
The 36 month data from the study support the lasting response and favorable safety and tolerability profile of Viaskin Peanut 250 µg in peanut-allergic children six to 11 years of age.
At the conclusion of the study, 61% of the children could tolerate 1,000mg (approximately four peanuts) or more cumulative reactive doses of peanut protein while 39% could tolerate 5,040mg (approximately 20 peanuts) or more.
Adverse reactions were limited to mild or moderate application-site reactions, the severity and frequency of which diminished with time. At no time during the study was epinephrine administration required for symptoms of anaphylaxis.
“It is encouraging to see that with continued treatment, the effect of Viaskin Peanut for children was sustained or increased throughout three years, while also maintaining a favorable and easily manageable safety profile,” said Dr Shreffler.
Phase III trials will commence later this year. The company is also in various stages of clinical trials for milk and egg versions of their therapy.
For a first-hand account from a parent of a child participating in the Viaskin Peanut trial, click here to see our friend Sharon Wong’s article on her blog, The Nut Free Wok.