Home Search
dbv - search results
If you're not happy with the results, please do another search
DBV, Developers of Patch Therapies for Allergies to Peanuts, Milk and Eggs, to Lay...
The company announced they would be laying off 200 employees in Q1.
FDA Has Questions About Viaskin Peanut Efficacy, Delays Meeting with DBV Technologies
The company warned the FDA's target date for their decision on approval could be delayed.
DBV Technologies Reports Positive Three-Year, Long Term Data from Phase III Open-Label Extension Study...
Patients demonstrated durable, long-term clinical benefit with an additional two years of treatment.
DBV Technologies Announces FDA Acceptance of BLA filing for Viaskin Peanut for the Treatment...
If approved, Viaskin Peanut would be the first and only epicutaneous immunotherapy indicated for this potentially life-threatening condition in children.
DBV Technologies Submits Biologics License Application to FDA for Viaskin Peanut for the Treatment...
This submission addresses the additional data needed on manufacturing procedures and quality controls which were communicated by the FDA in December.
DBV to Resubmit Application to FDA for Viaskin Peanut Approval in Q3 2019
Company allays concerns that BLA would not be resubmitted.
DBV Withdraws FDA Application for Viaskin Peanut with Plan to Resubmit
The company believes the additional information needed to support this filing is available without further clinical studies.
DBV Applies for FDA License to Sell Viaskin Peanut Patch Therapy
"This submission represents a significant step forward for those families living with peanut allergy."
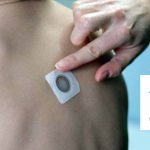
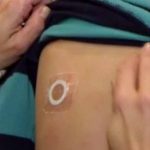
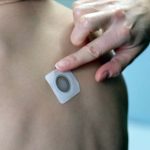
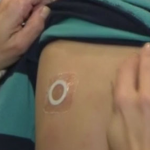
DBV Completes Phase III Study of Peanut Patch
The phase III trial was designed to assess the safety and routine clinical use of Viaskin Peanut.
Nestlé Partners with DBV to Develop Milk Allergy Test
The world's largest food business forays into allergy diagnostic services to help sell infant formula.