Home Search
dbv - search results
If you're not happy with the results, please do another search
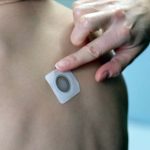
DBV Technologies Announces Positive 3-Year Results from EPITOPE Phase 3 Open-Label Extension Study
68.2% of subjects completed the oral food challenge (~12-14 peanut kernels) without meeting stopping criteria, compared to 30.7% at month 12.
DBV Confirms Alignment with FDA on Accelerated Approval Pathway for the Viaskin® Peanut Patch in...
Toddlers study on-track to initiate in 2Q 2025.
DBV Announces Positive Regulatory Updates for the Viaskin® Peanut Patch in the United States and...
Company to pursue an Accelerated Approval pathway for toddlers ages 1–3 years-old.
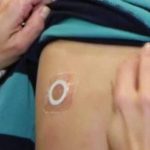
DBV Announces Promising 2-Year Results from Ongoing Viaskin “Peanut Patch” Toddler Study
81.3% of subjects who completed the oral food challenge reached an eliciting dose of ≥1,000 mg after 24 months of treatment.
DBV Outlines Path to Regulatory Approval of Viaskin Peanut Patch
Are Viaskin therapies finally on the horizon?
FDA Removes Hold Allowing DBV’s Pivotal Phase 3 Peanut Patch Trial to Commence
The phase 3 trial of the modified Viaskin Peanut patch has been cleared for commencement.
Private Law Firm to Investigate Claims on Behalf of Investors in DBV Technologies
It is unknown how this development will affect the ability of the company to eventually offer Viaskin Peanut as an FDA approved therapy.
FDA Issues Partial Hold on DBV Viaskin Peanut Patch Trial
Another delay for the long-awaited therapy.
DBV Begins Phase 3 Study of Redesigned Viaskin Peanut Patch in Children Ages 4-7
Viaskin is testing a new version of their patch designed to address the FDA's concerns.
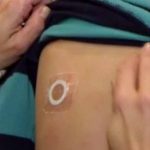
DBV Announces Positive Topline Results from Phase 3 Trial of Viaskin Peanut in Peanut-Allergic...
Viaskin Peanut demonstrated a statistically significant treatment effect with 67.0% of subjects meeting the treatment responder criteria after 12 months.