ATLANTA, GEORGIA (PRWEB) JULY 10, 2023
Moonlight Therapeutics, Inc., a biotechnology company developing an intracutaneous treatment for food allergy, announced it has received a multi-million dollar Fast-Track small business clinical trial grant to evaluate its treatment, MOON101, for peanut allergy in peanut allergic children and adults. The clinical trial grant was awarded by the National Institute of Allergy and Infectious Diseases (NIAID), a division of the National Institutes of Health (NIH). Moonlight has already completed a pre-IND meeting with the FDA on the first-in-human clinical trial for MOON101.
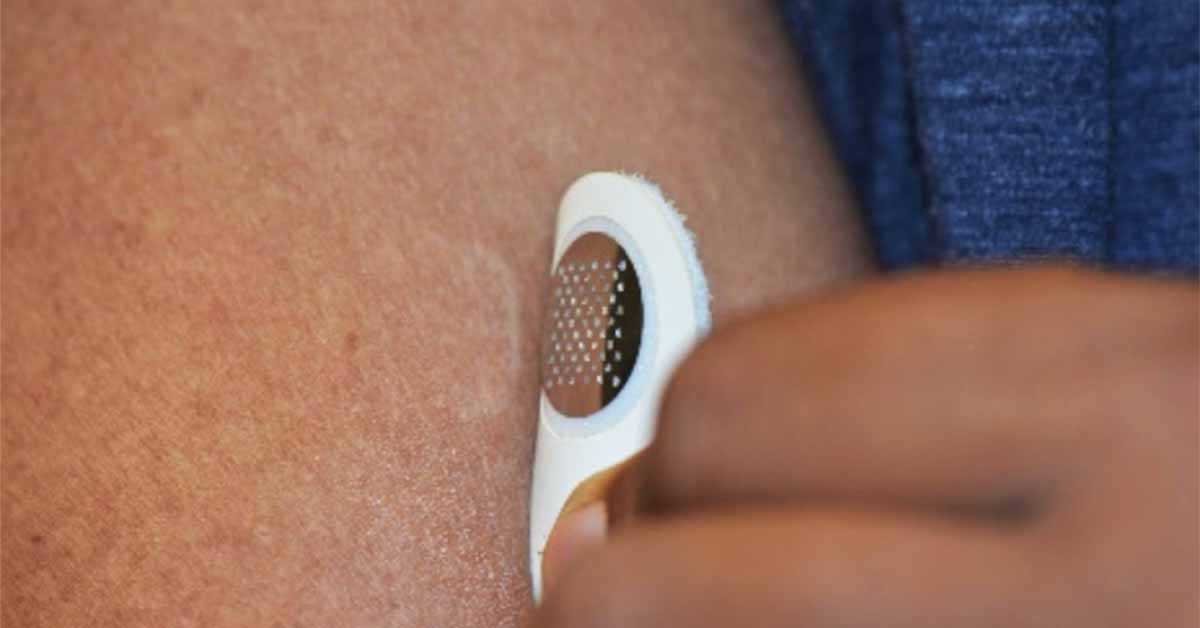
MOON101 uses Moonlight’s proprietary platform, a small, minimally invasive skin stamp, designed to break through the top-most layers of the skin barrier and deliver allergens to the skin’s immune cells in order to desensitize someone suffering from a food allergy. The dermal stamp is designed for at home self-administration. It is applied for only a few minutes and then removed. The approach has shown promising results in animal models of peanut and other allergies; data from these studies have been published in peer-reviewed journals.
This grant is the second small business grant the company has received from NIAID to develop MOON101 for the treatment of peanut allergy, bringing the company’s total non-dilutive funding to over $5 million. “This clinical trial grant represents further scientific validation of the company’s promising technology to address a large unmet need in the treatment of peanut and other food allergies,” said Samir Patel, Moonlight Therapeutics co-founder and CEO. “We were awarded this grant through a highly competitive review and it represents the meaningful progress we have made in advancing MOON101 towards the clinic.”
The clinical trial will be conducted in conjunction with several leading food allergy centers and clinical investigators in the United States. “There are over 6 million individuals in the United States alone with a peanut allergy and with only one FDA-approved therapy, there is still a large unmet need for patients and their families,” said Dr. Brian Vickery, who is the Director of the Food Allergy Center at Emory and Children’s Healthcare of Atlanta and serves as the lead clinical investigator on this grant. Dr. Vickery has been an investigator in food allergy clinical trials conducted at several stages of development including registration trials. “Clinical data from this grant will allow us to evaluate the effects of MOON101 directly on peanut allergic patients.”
“This award provides validation for the scientific approach Moonlight is taking and will facilitate the company’s entry into clinical trials,” added Dr. James Baker MD who is the founding Director of the Mary H. Weiser Food Allergy Center at University of Michigan and a former CEO of Food Allergy Research and Education (FARE), the national food allergy foundation.
Moonlight will be raising private funds to complement this award in order to accelerate the collection of the clinical data and development of MOON101.
About Moonlight Therapeutics
Moonlight Therapeutics is an early-stage biotechnology company based in Atlanta, Georgia. Moonlight is developing an intracutaneous allergen-specific immunotherapy platform for treating for food allergies. Food allergies are estimated to affect more than 30 million people in the United States. Moonlight has developed its proprietary platform, to break through the skin barrier and deliver allergens to immune cells in the top layer of the skin. This platform can be used to deliver individual or combinations of allergens to treat multiple allergies. This technology was invented at Texas Tech University and is supported by the Georgia Research Alliance. To learn more, visit moonlighttx.com.
NOTICE: This document contains certain forward-looking statements, including without limitation statements regarding Moonlight’s plans for pre-clinical and clinical studies, regulatory filings, investor returns and anticipated drug effects in human subjects. You are cautioned that such forward-looking statements are not guarantees of future performance and involve risks and uncertainties inherent in Moonlight’s business which could significantly affect expected results, including without limitation progress of drug development, ability to raise capital to fund drug development, clinical testing and regulatory approval, developments in raw material and personnel costs, and legislative, fiscal, and other regulatory measures. All forward-looking statements are qualified in their entirety by this cautionary statement, and Moonlight undertakes no obligation to revise or update any forward-looking statement to reflect events or circumstances after the issuance of this press release.